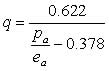Francis Massen, in response to the posts,
Comment On The Northeast Heat Wave
Further Information On The Role Of Water Vapor In Measuring Heat By Francis Massen
has graciously prepared a write up of how to compute the moist enthalpy of surface air. Francis websites include http://meteo.lcd.lu/; http://computarium.lcd.lu/; and http://bmb.lcd.lu/
Calculating Moist Enthalpy From Usual Meteorological Measurements By Francis Massen
Abstract: This short article shows how to compute the moist enthalpy from usual meteorological measurements of dry temperature, air pressure and relative humidity. The result is used to add a plot of moist air enthalpy to the other near-live graphs shown by meteoLCD, the meteorological station of the LCD, Diekirch, Luxembourg
1. Sensible heat of dry air
The sensible heat of dry air is defined as Ha = Cp*T [ref. 3] with Cp usually taken as 1.005 when Ha is given in [kJ/kg] and temperature T in [°C].
Here we will use for Cp the following expression, valid for temperatures higher than 0 °C and lower than 60 °C, as given by PADFIELD [ref.2]
Ha = 1.007*T – 0.026 0 °C < T < 60 °C [eq.1]
2. Heat content of water vapor at temperature T
The heat content of water vapor is the sum of the latent heat of vaporization and the sensible heat of water vapor:
Hv = q*( L + 1.84*T) [ref. TET] [eq.2]
Where L = heat of vaporization = 2501 kJ/kg at 0°C
and 1.84*T = sensible heat of water vapor in kJ/kg
The sensible heat term of eq.3 (1.84*T) is very often considered negligible and omitted.
Note:L is a function of temperature, becoming slightly smaller with increasing T; for values between 0°C and 50°C one can use the linear interpolation L(T) = 2502 – 2.378*T computed by the author from a table with enthalpy values given by YHCHEN [ref.4]: The linear fit is excellent with R2 = 0.9998.
Combining eq.2 with L(T) gives:
Hv = q*(2502 – 0.538*T) with Hv in kJ/kg and T in °C [eq.3]
3. Total enthalpy of moist air
Total enthalpy of moist air is the sum of Ha and Hv:
H = Ha + Hv = (1.007*T -0.026) + q*(2502 – 0.538*T) [eq.4]
with H in kJ/kg, T in °C and specific humidity q in kg/kg
The problem with this formula is that the specific humidity q is usually not measured by a standard meteorological equipment which commonly measures relative humidity.
4. Finding q from measured dry bulb temperature, relative humidity and atmospheric pressure
PIELKE [ref.3] and the AOMIP website [ref.1] give the following formula for the specific humidity q:
where ea = vapor pressure in [Pa] and pa = atmospheric pressure in [Pa].
Attention: pa is the true air pressure, not the barometric pressure reduced to sea level!
Dividing numerator and denominator by ea gives:
Relative humidity is the fraction of water vapor pressure to saturated water vapor pressure, usually multiplied by 100 to give a percent value:
RH = 100* ea/esat → ea = RH/100*esat
There are many different formulas relating esat to temperature. We will use the expression given in AOMIP [ref.1] and valid up to 40°C:
with saturated water vapor pressure esat in [Pa] and temperature T in °C.
Equations 4, 6 and 7 contain only T, RH and pa, which are parameters measured by practically every standard weather station. Together they can be used to calculate the enthalpy of moist air by a single (albeit unwieldy) formula:
This expression is valid for temperatures 0°C < T < 40°C. Units: H[kJ/kg], T[°C], pa[Pa]
5. A practical example
The author has used eq.8 in GNUPLOT to display near-live plots of the moist enthalpy at meteoLCD, Diekirch, Luxembourg (see http://meteo.lcd.lu/today_01.html). The following figure shows the situation for the week from 10 to 16th July 2010. Sensible heat is shown by the blue bottom curve; the difference between the upper red curve ( = moist enthalpy) and the blue curve corresponds to the latent heat.
Technisolve Software has a website with an online moist air calculator, which is very handy for a quick validation check of individual values: http://www.coolit.co.za/airstate/airmoistobject.htm

References
[1] AOMIP: Atmospheric Forcing Data – Humidity
http://efdl.cims.nyu.edu/project_aomip/forcing_data/atmosphere/humidity.html
[2] PADFIELD, Tim: Conservation Physics
http://www.conservationphysics.org/atmcalc/atmoclc1.php
[3] PIELKE, Roger, Sr., WOLTER, Klaus: The July 2005 Denver Heat Wave: How unusual was it ?. National Weather Digest, vol.31, no. 1, July 2007
http://pielkeclimatesciencesci.files.wordpress.com/2009/10/r-313.pdf
[4] TET (The Engineering Toolbox)
http://www.engineeringtoolbox.com/enthalpy-moist-air-d_683.html
[5] YHCHEN: Calculation of Enthalpy Changes